 
This molar mass calculator can only handle two bracket levels at a time. However, if we have 6. The total number of atoms in a substance can also be determined by using the relationship between grams, moles, and atoms. According to the periodic table, the atomic mass of aluminum is 26.98 amu, copper is 63.55 amu, and carbon is 12.01 amu.Since 1 amu is only 1.674 × 10 24 g, these masses would be way too small to measure on ordinary laboratory equipment. 
Tricalcium phosphate would be entered as Ca3(PO4)2. Multiply moles of Ca by the conversion factor (molar mass of calcium) 40.08 g Ca/ 1 mol Ca, which then allows the cancelation of moles, leaving grams of Ca. For example, calcium carbonate would be entered as CaCO3, not caco3. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. Lithium chloride is a solid which absorbs water to form a hydrate, LiCl.H 2 O. The chemical formula should be entered using standard format. The solid and the solution is separated and the supernatant solution is recycled for further evaporation. This calculator is a convenient tool for calculating the molar mass of chemical compounds in lieu of using a periodic table. These include consumption of pH adjustment chemicals for RO feedwater, solubilities of scale forming compounds in reverse osmosis systems, and cation rejection calculations using charge balance (meq/l) in nanofiltration systems. Many other calculations require conversion into moles. Knowing the desired concentration of ClO2, the system integrator can calculate the consumption of each of the reactants using the stoichiometric relationship:ĢNaClO2 + NaOCl + 2HCl ↔ 2ClO2 + H2O + 3NaCl For example, certain types of chlorine dioxide (ClO2) generators would use sodium hypochlorite (NaOCl), sodium chlorite (NaClO2) and hydrochloric acid (HCl). 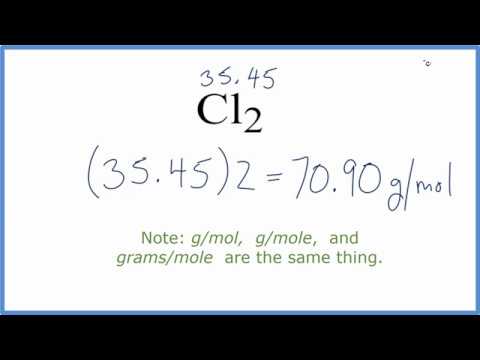
You can see that in Cl2, there are 2 Chlorine atoms. 1 Now, to calculate the molar mass of Cl2, you just have to add the molar mass of all the individual atoms that are present in Cl2. You can see that The molar mass of Chlorine is 35.45 g/mol. When calculating consumption of certain RO chemicals for reverse osmosis pretreatment or post-treatment, it is often necessary to convert into moles. So let’s look at the molar mass of Chlorine from the above periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
